Tanabe-Sugano diagrams are constructed from calculations based on ligand field theory and have been widely used in the analysis of absorption spectra of d 1 to d 9 ions. 
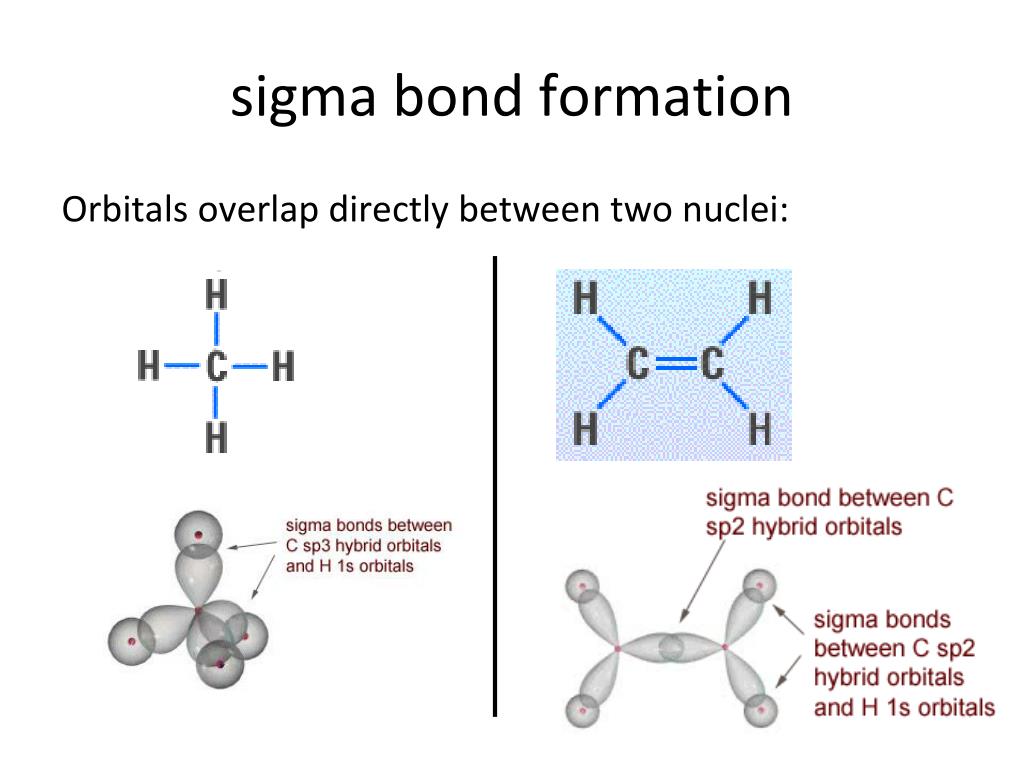
(b) Two of the hybrid orbitals on oxygen contain lone pairs, and the other two overlap with the 1s orbitals of hydrogen atoms to form the OH bonds in H2O. The electron density is concentrated along the bond axis. H sp hybridized carbon carbon atom shape linear bond angles. The overlapping extent of a sigma bond is greater than that of a pi bond The sigma bond which is a covalent bond is formed by headtohead overlap whereas the. Cyanides are released in nature via cyanogenic compounds and also produced by several microorganisms. 6: (a) A water molecule has four regions of electron density, so VSEPR theory predicts a tetrahedral arrangement of hybrid orbitals. Sigma and pi bonds All single bonds are referred to as ‘sigma’ bonds (-bonds). If we tried to add other non- hydrogen atoms, it would get too messy, as well. It carries a charge of -1 and is a conjugate base of hydrogen cyanide (HCN). It belongs to the cyano group and consists of carbon and a nitrogen atom having a triple bond. \): - A visible absorption spectrum of 3+. CN is known as cyanide which exists as a pseudohalide anion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
